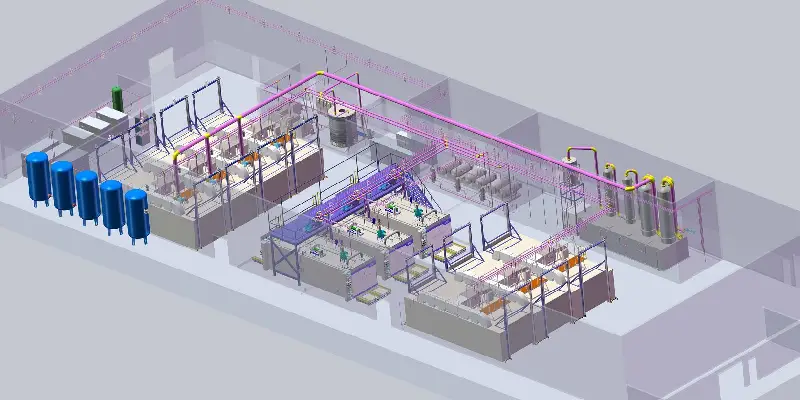
What Is The Importance Of Humidity When Using Eto Sterilization
Discover the critical importance of humidity in Ethylene Oxide (ETO) Sterilisation. Learn why maintaining 30%RH-80%RH is essential for effective gas penetration and microbial lethality. Explore the risks of low and high humidity, including condensation and package damage
What Is The Importance Of Humidity When Using Eto Sterilization Read More »