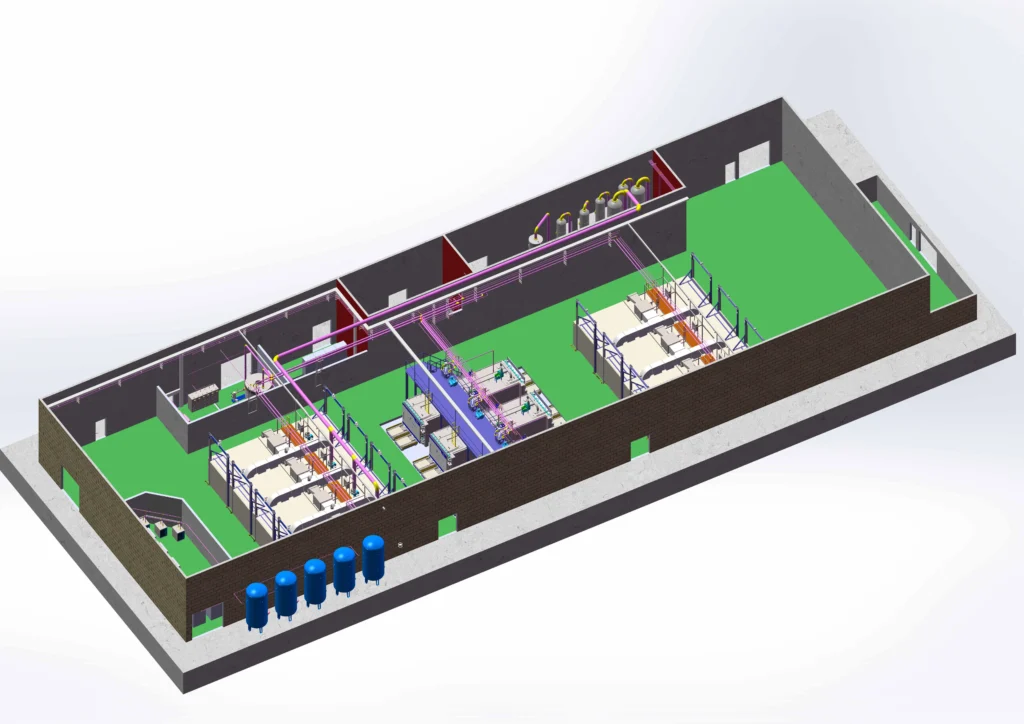
Choosing A Sterilization Contractor For Your Medical Device: 7 Key Questions To Ask
Choosing a sterilization contractor? Ask these 7 key questions about EO technology, material compatibility, chamber size, and turnaround times. Learn how BOCON in-house sterilization solves contractor delays.
Choosing A Sterilization Contractor For Your Medical Device: 7 Key Questions To Ask Read More »