Explosion Protection in Ethylene Oxide (環氧乙烷) 消毒
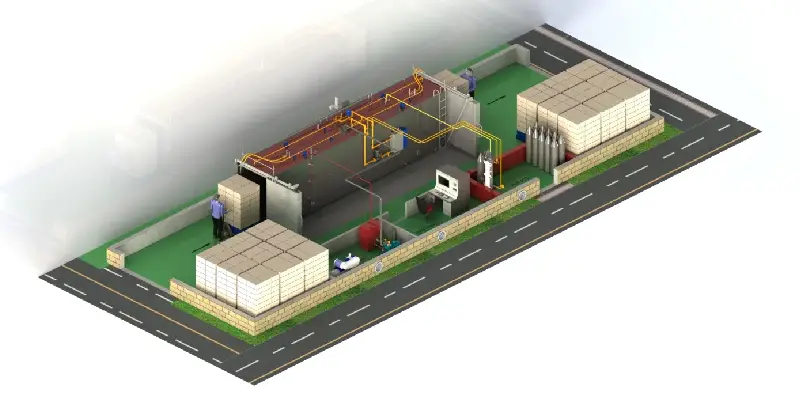
Explosion Protection for Ethylene Oxide (環氧乙烷) 消毒 | Technical Standards Guide Advanced Explosion Protection in Ethylene Oxide (環氧乙烷) Sterilization Processes Comprehensive Safety Analysis for Ethylene Oxide (環氧乙烷) Sterilization Facilities Expert Consultation for EO Sterilization 1. Explosion Concept and Risks in Ethylene Oxide (環氧乙烷) Sterilization An explosion occurs when the concentration of an explosive substance mixed […]
Explosion Protection in Ethylene Oxide (環氧乙烷) 消毒 閱讀更多 ”