手術縫合ETO滅菌器
Advanced Low-Temperature Solution for Synthetic & Bio-absorbable Sutures



Sterilization Method Comparison
| Method | Suitability for Sutures | Impact on Material Performance |
|---|---|---|
| 環氧乙烷 (ETO) | Industry Mainstream (高滲透率) | Minimal impact on tensile strength; shelf life up to 5 years. |
| Gamma Ray | Common Industrial Use | Risk of polymer degradation (e.g., PP, Polyester). |
| Steam/Boiling | Not Recommended | Significantly reduces strength; fibers may swell or embrittle. |
Tensile Strength
Our deep-vacuum cycles ensure zero structural damage, maintaining the critical strength needed for healing.
EO Residue Control
Strictly compliant with national standards (≤250μg/g) through optimized pulsating aeration cycles.
Moisture Content
For PGA/PLA sutures, our dry screw pump ensures moisture ≤0.05% to prevent premature hydrolysis.
Aeration & Degassing Performance
| Key Parameters | BOCON Dry Screw | Traditional System |
|---|---|---|
| Ultimate Vacuum | -101.27 千帕 | -97 千帕 |
| Hydrolysis Risk | Zero (Pure Dry) | High (Moisture Risk) |
| 遵守 | 國際標準化組織 11135 / 21 CFR Part 11 | Basic Standard |
Compact High-Efficiency Unit
Model: 1.5 CBM (1.5m³)
Specifically engineered for surgical suture manufacturers. Combines a small footprint with the deep-vacuum power of industrial-scale systems.

Professional FAQ
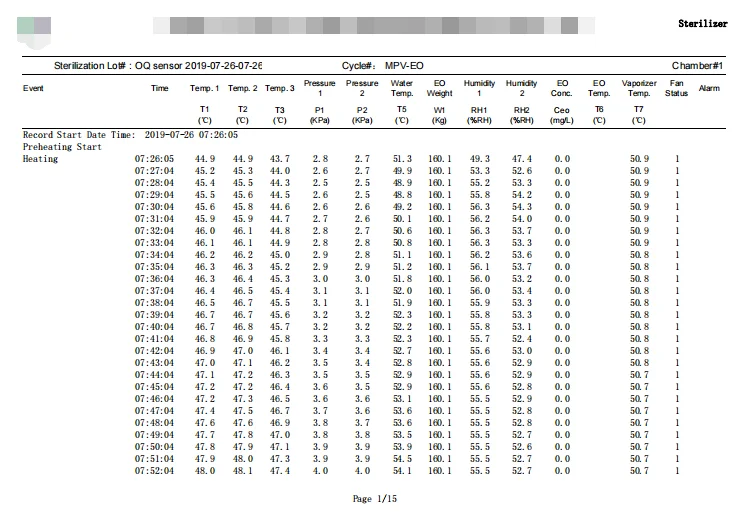
已驗證 滅菌週期 過程

載入和設定
打開裝載門, 裝載產品. 所有的門都關閉. 適當的參數設定. 開始循環.
加熱
加熱至52℃.
保持溫暖
系統將保溫一段時間 10 分分鐘.
預真空
根據用戶參數真空至-90kPa.
洩漏測試
持續時間是 13 分分鐘.
空氣到氮氣的替代
將腔室空氣更換為氮氣, 1-2 次.
加濕室
產品將被加濕至高達 55%RH.
濕度住宅
持續時間是 30 分分鐘.
N2 毯到腔室
EO 管路清洗和 EO 氣體注入室準備.
EO注入腔室
將 EO 氣體注入腔室以達到壓力 25 千帕.
氮氣蓋
流程是沖洗EO管道,讓所有EO進入腔室.
環氧乙烷住宅
它將堅持下去 2 小時和 10 分分鐘.
環氧乙烷去除
在真空下除去幾乎所有的 EO 氣體.
氮氣沖洗
為了安全起見,將 N2 氣體注入腔室以替換所有 EO 氣體.
空氣清洗
用空氣清洗產品, 經過空氣清洗過程後,EO 殘留量變得非常低.
通風
此流程與曝氣室功能相同, 確保 EO 殘留量更少.
循環完成
壓力上升至 -3.0 千帕~3.0千帕. 打開卸貨門,卸出產品. 列印歷史數據.
您值得信賴的全球 環氧乙烷滅菌器 夥伴
作為領先的 工業環氧乙烷滅菌器 製造商, BOCON提供的不僅是設備. 我們提供交鑰匙服務 ETO滅菌系統 支援 15+ 多年的卓越工程經驗. 從初始設計和 IQ/OQ/PQ 驗證到終身技術支持, we ensure your medical device sterilization process is safe, 合規的, 高效率.
全域安裝
由專家工程師現場設置和調試.
全面驗證
全面的 IQ/OQ/PQ 文件和支持.
24/7 支援
遠端診斷和快速備件交付.




評論
還沒有評論.