外科用縫合糸 ETO 滅菌器
Advanced Low-Temperature Solution for Synthetic & Bio-absorbable Sutures



Sterilization Method Comparison
| Method | Suitability for Sutures | Impact on Material Performance |
|---|---|---|
| エチレンオキサイド (ETO) | Industry Mainstream (High Penetration) | Minimal impact on tensile strength; shelf life up to 5 years. |
| Gamma Ray | Common Industrial Use | Risk of polymer degradation (e.g., PP, Polyester). |
| Steam/Boiling | Not Recommended | Significantly reduces strength; fibers may swell or embrittle. |
Tensile Strength
Our deep-vacuum cycles ensure zero structural damage, maintaining the critical strength needed for healing.
EO Residue Control
Strictly compliant with national standards (≤250μg/g) through optimized pulsating aeration cycles.
Moisture Content
For PGA/PLA sutures, our dry screw pump ensures moisture ≤0.05% to prevent premature hydrolysis.
Aeration & Degassing Performance
| Key Parameters | BOCON Dry Screw | Traditional System |
|---|---|---|
| Ultimate Vacuum | -101.27 kPa | -97 kPa |
| Hydrolysis Risk | Zero (Pure Dry) | High (Moisture Risk) |
| コンプライアンス | ISO 11135 / 21 CFR Part 11 | Basic Standard |
Compact High-Efficiency Unit
Model: 1.5 CBM (1.5m³)
Specifically engineered for surgical suture manufacturers. Combines a small footprint with the deep-vacuum power of industrial-scale systems.

Professional FAQ
Validated Sterilization Cycle Process
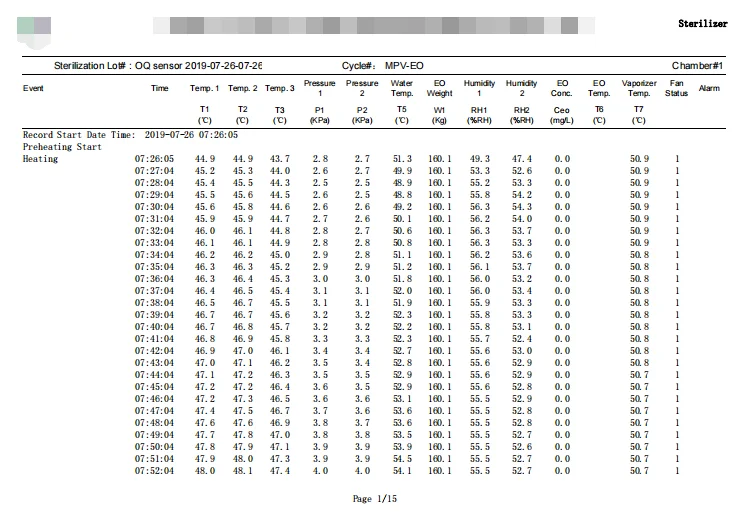

Loading & Setup
Open the loading door, load the products. All of the doors closing. Appropriate parameters setup. Start the cycle.
Heating
Heating up to 52℃.
Keep warm
The system will keep warm for 10 mins.
Pre-Vacuum
Vacuum to -90kPa according to the user parameters.
Leak testing
Duration is 13 mins.
Air to N2 replacement
Replacing the chamber air to N2 gas, 1-2 times.
Humidifying the chamber
The products will be humidified to up to 55%RH.
Humidity dwelling
Duration is 30 mins.
N2 blanket to the chamber
EO pipe washing and preparing the chamber for the EO gas injection.
EO injection to the chamber
The EO gas is injected into the chamber to reach pressure of 25 kPa.
N2 Cover
The process is to wash EO pipe and let all the EO go into the chamber.
EO Dwelling
It will hold on 2 hours and 10 mins.
EO removal
Removing almost all the EO gas out under vacuum.
N2 washing
Injection of N2 gas into the chamber to replace all the EO gas for safety.
Air washing
Washing the products with air, after the air washing process the EO residue becomes very low.
Aeration
This process has the same function with Aeration room, ensuring the EO residue much less.
Cycle Finished
Pressure goes up to -3.0 kPa~3.0 kPa. Open the unloading door and unload products. Print out history data.
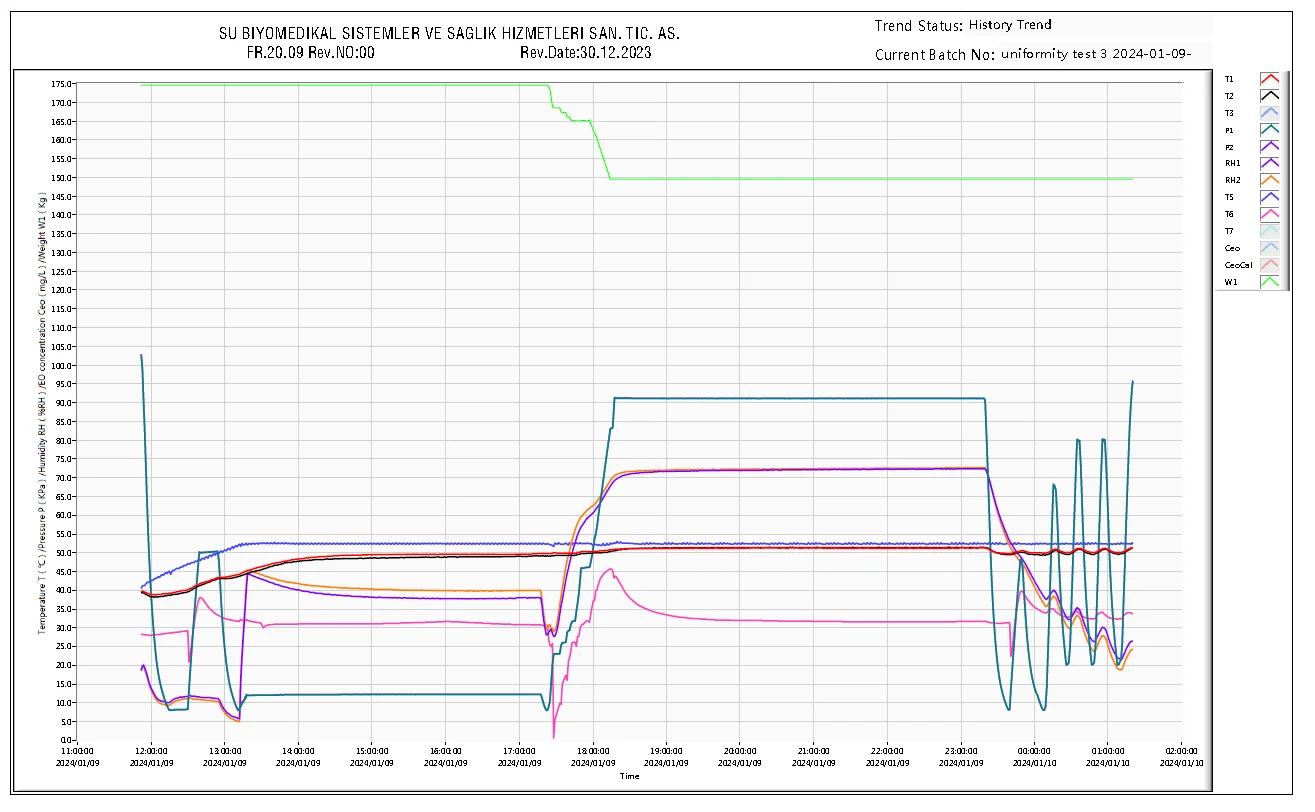
Your Trusted Global ETO Sterilizer パートナー
As a leading industrial ETO sterilizer manufacturer, BOCON provides more than just equipment. We deliver a turnkey ETO sterilization system backed by 15+ years of engineering excellence. From initial design and IQ/OQ/PQ validation to lifelong technical support, we ensure your medical device sterilization process is safe, compliant, and efficient.
Global Installation
On-site setup and commissioning by expert engineers.
Full Validation
Comprehensive IQ/OQ/PQ documentation and support.
24/7 サポート
Remote diagnostics and rapid spare parts delivery.
Ready to Deploy a Reliable
産業用 ETO 滅菌器 システム?
Whether you need a laboratory-scale unit or a large-scale industrial ETO sterilization system, our engineers are ready to provide a customized technical proposal.




レビュー
まだレビューはありません.