High-Performance Esterilizador industrial ETO & Systems
BOCON ETO Sterilizers are engineered for the rigorous demands of the global medical device industry. Our systems provide a fully automated, highly efficient, and 100% safe environment for the sterilization of heat and moisture-sensitive medical products.
From 1m³ laboratory units to 100m³ large-scale industrial chambers, we deliver more than just equipment—we provide a complete EO Sterilization System designed to meet ISO 11135 and CE estándares.

Unmatched Technical Advantages
5-Year Warranty
We provide a 5-year warranty for the entire machine, demonstrating absolute confidence in our equipment's stability.
Top-Tier Engineering
Led by Chief Engineer Mr. Sheng (15+ yrs experience), expert in high-rise automation and thermal power control.
Global Leadership
Preferred supplier for Medline, Cardinal, and Medtronic, holding 90%+ share among China's Top 3 manufacturers.
Advanced Control System
- Unrivaled Stability: Nuestras características de software 400+ auto-assessment functions and 120+ predictive alarms.
- Full Traceability: Designed to pass the most rigorous audits from Medtronic and Cardinal Health.
- Smart Integration: Standardized MES interfaces for cloud data backup and remote diagnostic capabilities.

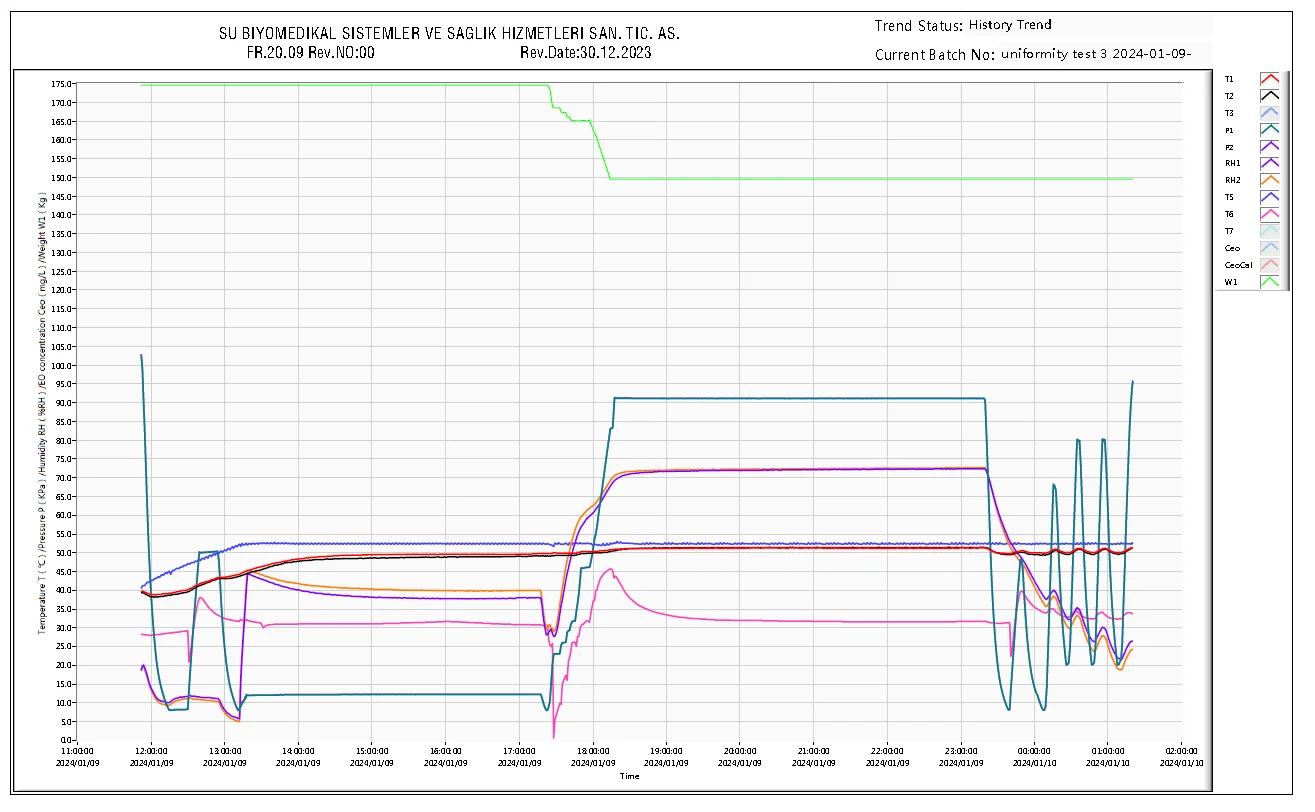
Fig 4.1: BCN-GE Series Automatic EO Control System Interface
Superior Craftsmanship
- Argon Shielded Welding: We use high-purity Argon protection welding to ensure structural longevity and vacuum integrity.
- Premium Components: Unlike competitors, we utilize proprietary Ex-sensors and high-performance burners for maximum safety.
- Quality over Price: Our higher price reflects meticulous design and industrial-grade hardware stability.

BOCON Industrial Welding Standard with Argon Protection
Proven Industrial Scale
- Turnkey Capability: We provide full installation, commissioning, and validation (IQ/OQ/PQ) services globally.
- Continuous Choice: Leading manufacturers repeatedly return to BOCON after testing other domestic alternatives.
- R&D Investment: 2-3 million RMB annual investment with 28 international and domestic patents.

Full-scale Industrial ETO Sterilization System Implementation
BOCON ETO Sterilizer Specifications
Global Standards Compliance: ISO 11135, EN 1422, CE, ISO 13485. Full Support for FAT, SE SENTÓ, DQ, coeficiente intelectual, OQ, PQ validation.
Technical Support
Need custom dimensions or automated conveyor loading systems for your EO Sterilization line?
CONSULT AN ENGINEER →Validated Sterilization Cycle Process

Loading & Setup
Open the loading door, load the products. All of the doors closing. Appropriate parameters setup. Start the cycle.
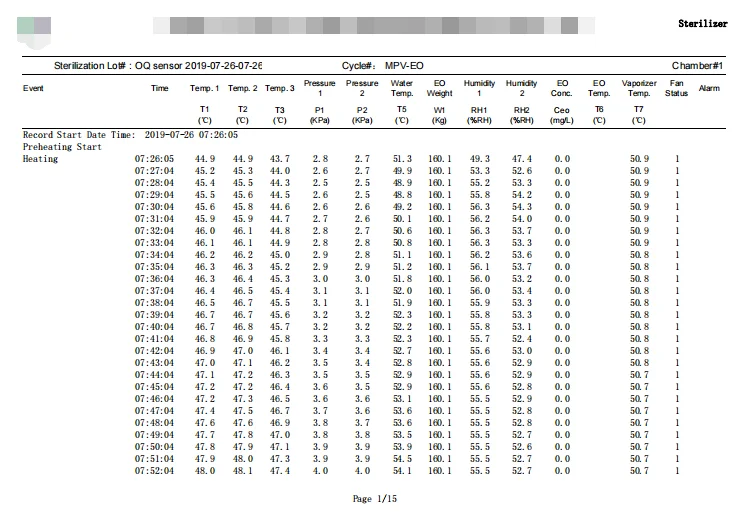
Heating
Heating up to 52℃.
Keep warm
The system will keep warm for 10 mins.
Pre-Vacuum
Vacuum to -90kPa according to the user parameters.
Leak testing
Duration is 13 mins.
Air to N2 replacement
Replacing the chamber air to N2 gas, 1-2 times.
Humidifying the chamber
The products will be humidified to up to 55%RH.
Humidity dwelling
Duration is 30 mins.
N2 blanket to the chamber
EO pipe washing and preparing the chamber for the EO gas injection.
EO injection to the chamber
The EO gas is injected into the chamber to reach pressure of 25 kPa.
N2 Cover
The process is to wash EO pipe and let all the EO go into the chamber.
EO Dwelling
It will hold on 2 hours and 10 mins.
EO removal
Removing almost all the EO gas out under vacuum.
N2 washing
Injection of N2 gas into the chamber to replace all the EO gas for safety.
Air washing
Washing the products with air, after the air washing process the EO residue becomes very low.
Aeration
This process has the same function with Aeration room, ensuring the EO residue much less.
Cycle Finished
Pressure goes up to -3.0 kPa~3.0 kPa. Open the unloading door and unload products. Print out history data.
Your Trusted Global ETO Sterilizer Pareja
As a leading industrial ETO sterilizer manufacturer, BOCON provides more than just equipment. We deliver a turnkey ETO sterilization system backed by 15+ years of engineering excellence. From initial design and IQ/OQ/PQ validation to lifelong technical support, we ensure your medical device sterilization process is safe, compliant, and efficient.
Global Installation
On-site setup and commissioning by expert engineers.
Full Validation
Comprehensive IQ/OQ/PQ documentation and support.
24/7 Support
Remote diagnostics and rapid spare parts delivery.
ETO Sterilizer FAQ
Frequently asked questions about our industrial sterilization solutions.
- High Penetration: Perfect for large volume sterilization because of the penetration property of EO gas.
- Final Packaging: Sterilize after final packing to avoid secondary pollution.
- Material Safety: Low temperature processing maintains the original characteristics of the product.
- Global Trust: Long history of safe and effective use with ~50,000 medical devices validated.
- Low Maintenance: Designed for industrial scale with low operation and maintenance costs.
Our Industrial ETO Sterilizer solutions are suitable for a wide range of products. For specific consultations, please contact us.
- Endoscopes: Bronchoscopes, cystoscopes, esophagoscopes, mediastinoscopes, etc.
- Plastic & Rubber: Gloves, syringes, infusion devices, catheters, and bags.
- Instruments: Pacemakers, artificial bones, sutures, bone plates, and medical electric drills.
- Anesthetic Vessels: Endotracheal tubes, masks, oxygen tents, and atomizers.
- Textiles: Surgical gowns, caps, bedding, gauze, and bandages.
Ready to Deploy a Reliable
Esterilizador industrial ETO System?
Whether you need a laboratory-scale unit or a large-scale industrial ETO sterilization system, our engineers are ready to provide a customized technical proposal.




Reviews
There are no reviews yet.